Classical ‘Philadelphia negative’ myeloproliferative neoplasms (MPNs) are clonal haematopoietic disorders and include essential thrombocythaemia (ET), polycythaemia vera (PV) and myelofibrosis (MF). MPNs have a heterogenous phenotype with an inherent risk of thromboembolic and haemorrhagic complications, and risk of blastic transformation. MPNs are commonly diagnosed in the 6 th decade or later, although up to 20% of patients are diagnosed < 40 years. Current risk stratification and treatment guidance, however, is frequently extrapolated from older cohorts.
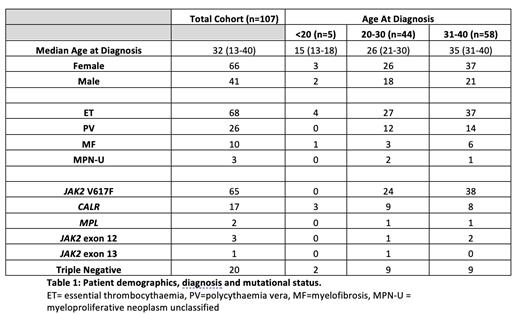
Here, we present a comprehensive retrospective analysis of 107 MPN adolescent and young adult (AYA) patients diagnosed <40 years, representing 19.6% of the total outpatient MPN cohort at University College London Hospital. Median follow up was 78 months (range (r), 1 - 507). Table 1 displays clinicopathological details of the entire cohort. Median age at diagnosis was 32 years (r,13-40). There was a female preponderance (66%); 57.9% of patients were Caucasian. Most patients had a diagnosis of ET (57.9%), with 24.3% PV and 9.3% MF, respectively. The most common driver mutation was JAK2 V617F, observed in 60.7%, followed by CALR (15.9%) and MPL (1.9%); 17% of patients were classified as triple negative (TN). Where data was available (n=27), median variant allelic frequency (VAF) for JAK2 V617F was 19.7% (r, 4-75%) and for CALR 43% (r, 5.7 - 53). 9/107 (8.4%) of patients had additional pathogenic gene abnormalities, most commonly variants of NRAS (22.2%), TET2 (22.2%) and ASXL1 (22.2%).
We observed a high thrombosis rate of 19.6% (21/107); the majority had a diagnosis of ET (61.9%).. In 80% of such cases, this was the heralding event leading to the MPN diagnosis. Venous thrombosis was more frequent than arterial thrombosis (68% vs 33%). In particular, there was a disproportionate number of venous thromboses at unusual sites including cerebral venous sinus and splanchnic venous thromboses, representing 40% and 33% of total events, respectively. Only 3 (2.8%) patients were found to have a co-existent thrombophilia. Younger age at diagnosis appeared to associate with a higher rate of thrombosis (60% in patients aged <20, 18.2% in patients aged 20-30, and 17.2% in patients aged 31-40). Thrombotic events were most common in those with a JAK2V617 mutation (23.4%), followed by those who were TN (20.0%). Only 5.9% of CALR mutated cases had a thrombotic event. In terms of cardiovascular risk factors, 13.0% (14/107) and 6.5% (7/107) of patients had diagnoses of hypertension and type 2 diabetes, respectively.
Haemorrhagic events were reported in 8.4% (9/107) of patients; 3.7% (4/107) had grade 3 or 4 bleeding complications, requiring transfusion and/or surgical intervention. All who experienced haemorrhagic events were on treatment with an anti-platelet or anti-coagulant agent. 4.7% (5/107) were diagnosed with acquired von Willebrand's disease, of whom one experienced a grade 3 haemorrhagic event. No patients transformed during follow up. 33 patients reported pregnancies, with 7/33 (21%) reporting antenatal or perinatal medical complications.
Over 50% (55/107) of patients were commenced on cytoreduction. Most common agents employed were pegylated interferon (45.5%; 25/55) and hydroxycarbamide (43.6%%; 24/55). No patients required combination therapy. 92/107 (86%) patients were treated with an antiplatelet agent and 9/107 (8.4%) were on anti-coagulant treatment with the majority (88.8%) receiving warfarin rather than a DOAC.
AYA MPN patients have unique characteristics and heterogeneous clinico-biological features differentiating them from older MPN cohorts. Our data highlights a female preponderance, higher incidence of venous thrombosis (particularly atypical sites), and lower incidence of arterial thrombosis. Whilst traditionally younger patients are considered to have ‘low risk’ disease, this real-world study highlights a significant thrombosis rate of ~ 20% which may have associated morbidity considering the relatively longer duration of the disease course and need for cytoreductive therapy. Specific AYA MPN directed risk stratification and therapeutic algorithms are required to inform holistic and age-appropriate patient management.
Disclosures
McLornan:UK ALL RIC TRIAL - DSM board: Other: participation on a data safety monitoring board or advisory board; Novartis: Honoraria; EBMT Scientific Council Member: Other: Chair of EBMT CMWP; Abbvie: Honoraria; Jazz Pharma: Honoraria; Imago Biosciences: Research Funding. Lambert:Kite-Gilead: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria; Blueprint: Membership on an entity's Board of Directors or advisory committees; Takeda: Honoraria. Wilson:Imago BioSciences, Inc., a subsidiary of Merck & Co., Inc., Rahway, NJ, USA: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal